If you are considering in vitro fertilization (IVF), you may worry about the health of your future child and the emotional toll of repeated unsuccessful cycles. Preimplantation Genetic Testing (PGT) offers a way to screen embryos for genetic abnormalities before transfer, aiming to increase your chances of a healthy pregnancy and reduce the risk of passing on inherited conditions. In the United States, more than a third of fertility centers now use PGT, reflecting its growing role in helping families achieve their goals safely and confidently.
Your concerns are valid: research shows that even in younger women, up to 40% of embryos can have chromosomal abnormalities, which can lead to failed implantation or miscarriage. PGT helps identify embryos with the best chance of developing into a healthy baby, improving pregnancy rates per transfer, especially for women over 35 or those with a history of genetic disease. While no technology can guarantee success, PGT represents a significant step forward in making your fertility journey more predictable and compassionate.
- What is preimplantation genetic testing and how has the technology evolved?
- How does PGT fit into the IVF workflow, step by step?
- Which PGT type solves which genetic problem?
- Does PGT really boost IVF success and cut miscarriage rates?
- What cutting-edge technologies are pushing PGT forward right now?
- Who benefits most from PGT—and when might it be unnecessary?
- How should you handle mosaic embryos and inconclusive results?
- What risks, limitations, and ethical debates come with selecting embryos by genetics?
- How much does PGT cost, and is it cost-effective?
- Which key questions should you ask your clinic before choosing PGT?
- Where is PGT headed over the next five years?
- Your Top Questions, Answered
- Final Thoughts
- References
What is preimplantation genetic testing and how has the technology evolved?
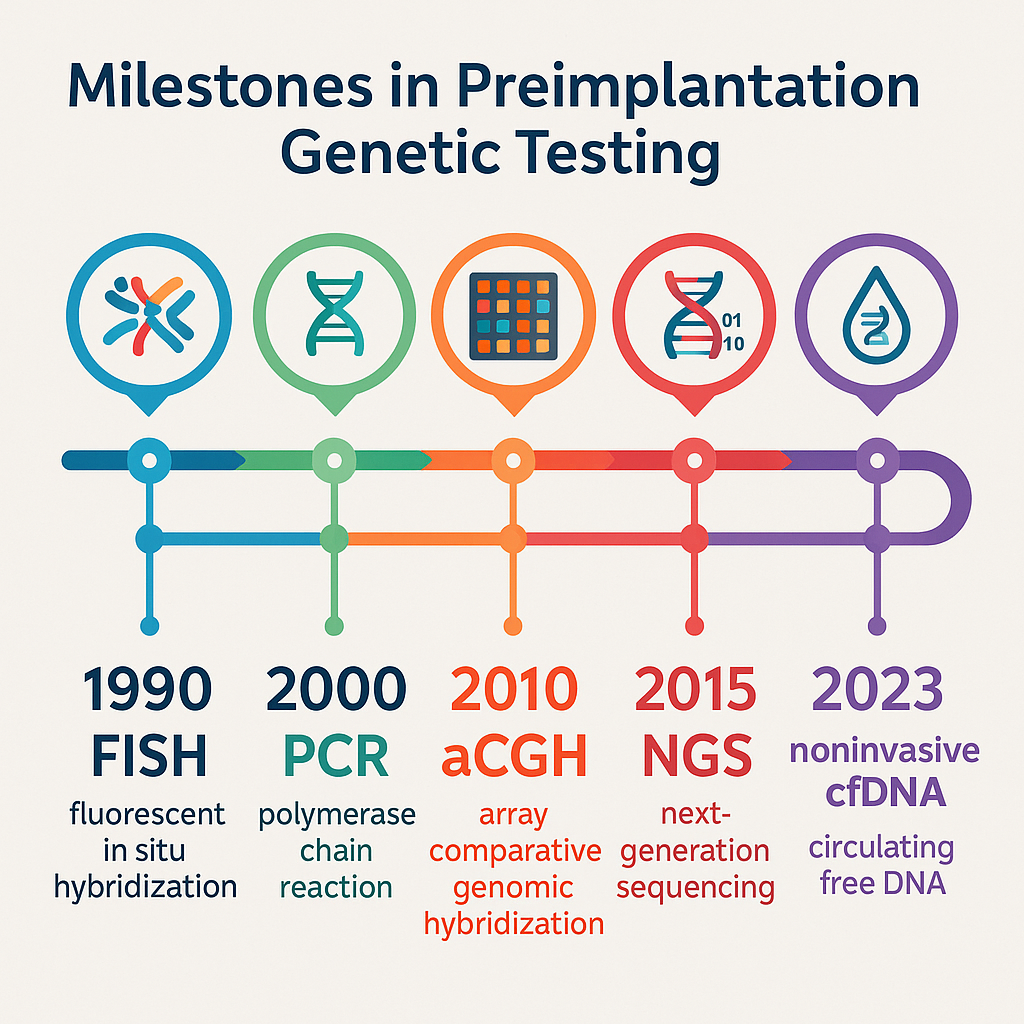
Preimplantation genetic testing (PGT) is a laboratory technique used during in vitro fertilization (IVF) to analyze embryos for genetic abnormalities before transfer to the uterus. The goal is to select embryos with the best chance of resulting in a healthy pregnancy and to reduce the risk of inherited genetic disorders or chromosomal issues. PGT began with methods like single-cell PCR and fluorescent in situ hybridization, which could only test for a limited number of conditions or chromosomes. Over the past decade, the technology has advanced rapidly, moving to whole genome amplification, single nucleotide polymorphism (SNP) arrays, and next-generation sequencing (NGS), which allow for comprehensive screening of all chromosomes and many genetic diseases in a single test.
Recent innovations include non-invasive techniques that analyze cell-free DNA from the culture medium, reducing the risk of harming embryos during biopsy, though these methods are still being refined for accuracy and consistency. The integration of advanced bioinformatics and artificial intelligence is further improving the interpretation of complex genetic data, making PGT more efficient and accessible. Today, PGT can screen for monogenic disorders, chromosomal aneuploidies, and even help with HLA matching for certain medical needs. As the technology continues to evolve, it is becoming more reliable, faster, and less invasive, offering you greater confidence and more options in your fertility journey.
Curious how algorithms judge embryo images? Explore AI in IVF.
How does PGT fit into the IVF workflow, step by step?
Preimplantation genetic testing (PGT) is carefully integrated into the in vitro fertilization (IVF) process to help you select embryos with the highest chance of leading to a healthy pregnancy.
Ovarian stimulation: You receive hormone medications to stimulate your ovaries, encouraging the development of multiple eggs in one cycle.
Egg retrieval: When your eggs are mature, a doctor collects them from your ovaries using a minor surgical procedure.
Fertilization: The retrieved eggs are combined with sperm in the laboratory to create embryos.
Embryo culture: Embryos are grown in a controlled environment for several days, usually until they reach the blastocyst stage (day 5 or 6).
Embryo biopsy: A few cells are carefully removed from each viable embryo, typically from the outer layer (trophectoderm), without harming the embryo’s ability to develop.
Genetic analysis: The biopsied cells are sent to a genetics lab, where advanced techniques like next-generation sequencing (NGS) or comparative genomic hybridization (CGH) screen for chromosomal abnormalities or specific genetic conditions.
Embryo freezing: While awaiting test results, embryos are usually frozen (cryopreserved) to maintain their quality.
Results review: Your care team discusses the genetic findings with you, identifying which embryos are chromosomally normal (euploid) or free of targeted genetic disorders.
Embryo selection and transfer: You and your doctor select the healthiest embryo for transfer into your uterus in a future cycle, aiming to maximize your chance of a healthy pregnancy.
Remaining embryos: Any additional healthy embryos can be kept frozen for possible future use.
You and your doctor then select the healthiest embryo for transfer in a future cycle, aiming to maximize your chances of a successful pregnancy and reduce the risk of miscarriage or inherited disease. This step-by-step approach allows for informed, compassionate decision-making throughout your IVF journey.
Want to peek inside a modern lab? Take a VR IVF Lab Tour.
Which PGT type solves which genetic problem?
Preimplantation genetic testing (PGT) includes several types, each designed to address specific genetic problems in embryos created through IVF. PGT-A (Preimplantation Genetic Testing for Aneuploidy) screens embryos for abnormal numbers of chromosomes, helping to prevent conditions like Down syndrome and reduce the risk of miscarriage or failed implantation. PGT-M (Preimplantation Genetic Testing for Monogenic Disorders) targets single-gene diseases such as cystic fibrosis, sickle cell anemia, or Huntington’s disease, ensuring embryos do not inherit a known family mutation. PGT-SR (Preimplantation Genetic Testing for Structural Rearrangements) detects chromosomal structural changes, like translocations or inversions, which can cause infertility, miscarriage, or genetic disorders in offspring 1.
Emerging technologies now allow for simultaneous testing of aneuploidy, monogenic disorders, and structural rearrangements on a single platform, streamlining the process and increasing accuracy. Additionally, PGT-P (Preimplantation Genetic Testing for Polygenic Disorders) is being developed to estimate the risk of complex diseases influenced by multiple genes, such as type 1 diabetes or hypothyroidism, though this is still experimental. By matching the right PGT type to your specific genetic concern, you and your care team can make informed decisions to maximize the chance of a healthy pregnancy and child.
Does PGT really boost IVF success and cut miscarriage rates?
Preimplantation genetic testing (PGT), especially PGT-A for aneuploidy, can improve certain IVF outcomes, but its benefits depend on your individual situation. For people with recurrent pregnancy loss or repeated implantation failure, PGT-A is linked to higher live birth rates and lower miscarriage rates, helping to identify embryos with the best chance of success. In women over 37, PGT-A may increase the chances of pregnancy and live birth per embryo transfer. However, this advantage may not persist when looking at outcomes per IVF cycle or retrieval. For younger women or those with a good prognosis, PGT-A does not consistently boost live birth rates or reduce miscarriages compared to standard IVF.
PGT-A is most effective for those with specific indications, such as advanced maternal age, recurrent miscarriage, or known chromosomal issues, rather than as a universal solution for all IVF patients. While PGT-A can lower the risk of transferring embryos with chromosomal abnormalities, other factors like embryo quality, uterine health, and lifestyle also play a significant role in IVF success and miscarriage risk. In summary, PGT can help reduce miscarriage rates and improve success for certain groups, but it is not a guaranteed way to boost IVF outcomes for everyone. Your care team can help determine if PGT is right for your unique fertility journey.
Pairing PGT data with predictive analytics can fine-tune your individual odds.
What cutting-edge technologies are pushing PGT forward right now?
Cutting-edge technologies are rapidly advancing preimplantation genetic testing (PGT), making it more comprehensive, accurate, and informative. Next-generation sequencing (NGS) has become the backbone of modern PGT, enabling high-resolution analysis of all chromosomes, detection of mosaicism, and identification of small segmental imbalances that were previously undetectable. New genotyping methods now allow for parallel qualitative and quantitative DNA analysis. This can uncover subtle genetic variations, meiotic recombination events, and even help fingerprint embryos to prevent lab errors or contamination. Improved bioinformatics and artificial intelligence are enhancing the interpretation of complex genetic data, leading to more reliable embryo selection and standardized reporting.
Non-invasive PGT approaches, such as analyzing cell-free DNA from the embryo’s culture medium, are being developed to reduce the risks associated with embryo biopsy, though these methods are still being validated for clinical use. There is also a push toward universal PGT platforms that can simultaneously test for aneuploidy, monogenic disorders, and structural rearrangements from a single biopsy, streamlining the process and expanding its clinical utility. As these technologies evolve, they promise to improve the accuracy of detecting de novo mutations and complex genetic traits, moving PGT beyond just aneuploidy screening and toward a more holistic assessment of embryo health 3.
Who benefits most from PGT—and when might it be unnecessary?
PGT (preimplantation genetic testing) offers the most benefit to specific groups undergoing IVF, particularly women of advanced maternal age (typically over 35 or 38). Those with a history of recurrent pregnancy loss, recurrent implantation failure, or certain high-risk infertility factors such as severe male infertility or uterine adhesions. In these populations, PGT; especially PGT-A for aneuploidy, can increase live birth rates per embryo transfer, reduce miscarriage rates, and improve the chances of a healthy pregnancy by helping select embryos with the correct number of chromosomes.
For women under 35 or 37 years old, especially those undergoing their first IVF cycle and without a history of pregnancy complications, PGT-A does not significantly improve live birth or ongoing pregnancy rates compared to standard embryo selection based on appearance alone, and may add unnecessary cost and complexity.
PGT is generally unnecessary for younger women with good ovarian reserve and no history of repeated IVF failure or miscarriage, as their embryos are less likely to have chromosomal abnormalities and standard selection methods are usually sufficient. Additionally, PGT may not increase the overall cumulative live birth rate per egg retrieval cycle, though it can improve outcomes per transfer in select groups. Ultimately, the decision to use PGT should be individualized, weighing potential benefits against costs, risks, and personal reproductive history.
Wearable hormone trackers—see Fertility Wearables—can help predict how many eggs you might produce for testing.
How should you handle mosaic embryos and inconclusive results?
Managing mosaic embryos and inconclusive PGT results requires careful, individualized decision-making and thorough patient counseling. Mosaic embryos; those with a mix of normal and abnormal cells, can sometimes result in healthy pregnancies. This is especially when the proportion of abnormal cells is low (typically less than 50%), with recent evidence showing live birth rates similar to fully normal (euploid) embryos in these cases. Current guidelines recommend prioritizing the transfer of euploid embryos first, but if none are available, low-level mosaic embryos may be considered. This is particularly for patients with limited options, such as older women or those with no other embryos for transfer. High-level or complex mosaic embryos are associated with lower implantation and higher miscarriage rates, so their transfer should be approached with caution and only after detailed discussion of risks.
Inconclusive PGT results, where the test cannot clearly determine the embryo’s chromosomal status, should prompt a review of technical factors and may warrant repeat testing or prioritizing other embryos if available. All patients considering mosaic embryo transfer should receive genetic counseling about the potential for reduced success rates and the importance of prenatal testing if pregnancy occurs. Laboratories and clinics are encouraged to validate their reporting thresholds for mosaicism and to develop clear protocols for managing these cases. Ultimately, decisions should be made collaboratively between patients and clinicians, weighing the risks, benefits, and available alternatives.
What risks, limitations, and ethical debates come with selecting embryos by genetics?
Risks: Selecting embryos by genetics, especially using polygenic risk scores (PGT-P), carries several risks. The predictive power of these scores is limited, meaning that the expected health or trait benefits for the child are often small and uncertain, with wide confidence intervals and a real possibility of unintended consequences, such as inadvertently selecting for adverse traits or reducing the overall chance of pregnancy by discarding viable embryos. There is also a risk of unnecessary IVF treatments, patient confusion due to complex or poorly understood results, and the psychological burden of choice overload . Additionally, technical variability in how scores are calculated can lead to inconsistent or unreliable results, further increasing the risk of error.
Limitations: The main limitations include the low accuracy and limited clinical utility of current polygenic risk scores, especially for complex traits like intelligence or height, where the actual benefit from selection is minimal, often just a few IQ points or centimeters in height, with large uncertainty. The effectiveness of genetic selection is further constrained by the small number of embryos typically available in IVF cycles, which limits the potential for meaningful selection. Most genetic risk scores are less accurate for people of non-European ancestry, raising concerns about equity and generalizability. Technical challenges, such as differences in scoring methods and random fluctuations in score construction, can also affect which embryo is selected, making the process less reliable.
Ethical Debates: Ethical concerns are significant and multifaceted. There is widespread debate about the acceptability of selecting embryos for non-disease traits, such as intelligence or physical appearance, and fears about the rise of “designer babies”. This technology could exacerbate social inequalities, stigmatize disability, and overemphasize genetic factors at the expense of environmental and social influences . The use of opaque or “black box” AI models in embryo selection raises issues of transparency, accountability, and potential bias. There are also concerns about unequal access to these technologies, especially for people of non-European ancestry, and the risk of increasing pressure on parents to select for certain traits. Many experts recommend that, given these unresolved ethical and societal questions, genetic embryo selection should be limited to research settings until its broader impacts are better understood.
Ultimately, these debates highlight the need for robust regulation, transparent counseling, and ongoing ethical reflection as genetic embryo selection advances.
Broader tech comparisons appear in Smart Thermometers: Best Picks for Easy Tracking.
How much does PGT cost, and is it cost-effective?
The cost of preimplantation genetic testing (PGT) varies widely depending on the country, clinic, and type of test, but it typically adds several thousand dollars (or equivalent) to the cost of an IVF cycle, with estimates ranging from about $4,000 to over $10,000 per cycle in many settings. Regarding cost-effectiveness, PGT-A (for aneuploidy) is generally not considered cost-effective for routine use in the general IVF population, as it increases the cost per live birth and does not significantly improve cumulative live birth rates, especially in younger women or those with only one or two embryos.
However, PGT can be cost-effective for specific groups, such as older women, those with recurrent pregnancy loss, or couples at risk of transmitting serious genetic diseases (using PGT-M), where it can prevent costly and life-altering conditions in offspring. For example, PGT-M to prevent transmission of BRCA mutations or sickle cell disease has been shown to be cost-effective compared to the long-term healthcare costs of managing these conditions. Newer, more targeted sequencing technologies may further reduce costs and improve accessibility in the future. Ultimately, the cost-effectiveness of PGT depends on individual patient factors, the specific genetic risk, and local healthcare economics, and it is not universally recommended for all IVF patients.
Budget apps listed in Top Fertility Apps 2025 can help track expenses.
Which key questions should you ask your clinic before choosing PGT?
Here are key questions to ask your clinic before choosing preimplantation genetic testing (PGT):
- What type of PGT is being recommended for my situation, and why?
- What are the clinic’s success rates with PGT, including implantation and live birth rates?
- What are the total costs involved, and does insurance or financial assistance cover any part of it?
- Which laboratory will perform the testing, and how might their methods impact results?
- What are the chances of false positives, inconclusive, or failed results?
- What are the risks and limitations of PGT in my specific case?
- What happens to embryos that are not selected or are found to have genetic abnormalities?
- What emotional and psychological support is available during the process?
- What counseling services are offered to help me understand the results and make decisions?
- Are there any ethical or legal considerations I should be aware of regarding PGT?
Where is PGT headed over the next five years?
Over the next five years, preimplantation genetic testing (PGT) is expected to become more accurate, less invasive, and more comprehensive. Advances in sequencing technologies, such as third-generation and long-read sequencing, will allow for more detailed and cost-effective analysis of embryos, enabling simultaneous detection of chromosomal abnormalities, single-gene disorders, and structural rearrangements in a single test.
Non-invasive PGT methods, which analyze cell-free DNA from spent embryo culture medium instead of embryo biopsy, are being developed and may reduce risks to embryos and improve patient acceptance, though further validation is needed before routine clinical use. The integration of next-generation sequencing and improved bioinformatics will enhance diagnostic accuracy and may help address challenges like mosaicism and variants of unknown significance.
As costs decrease and technology becomes more accessible, PGT may be offered more widely, but the growing complexity of genetic data will require better genetic counseling and clear regulatory guidelines . Overall, the future of PGT points toward more personalized, safer, and efficient embryo selection, but ongoing research and careful oversight will be essential to ensure clinical value and address ethical, legal, and societal concerns .
Combine these with immersive anatomy tools such as 3D Uterus Models for a truly futuristic fertility plan.
Your Top Questions, Answered
Does PGT guarantee a healthy baby?
No, PGT cannot guarantee a healthy baby. While it reduces the risk of certain genetic diseases or chromosomal abnormalities, it cannot detect all possible conditions, and some issues may arise that are not screened for or are undetectable at the embryo stage.
Is embryo biopsy safe?
Embryo biopsy is generally considered safe and is widely used, but it is an invasive procedure that carries a small risk of harming the embryo or affecting its viability. The risk is lower with modern techniques, especially when performed at the blastocyst stage, but concerns remain, particularly regarding mosaicism and technical variability.
Can PGT detect every disease?
No, PGT cannot detect every disease. It is designed to identify specific genetic mutations or chromosomal abnormalities, but it cannot screen for all possible genetic or developmental problems, especially those that may arise later in pregnancy or after birth.
What happens to embryos flagged as abnormal?
Embryos identified as having genetic abnormalities are typically not selected for transfer. Depending on clinic policy and patient preferences, they may be discarded, frozen for future research, or, in some cases, donated for scientific study.
Is non-invasive PGT available now?
Non-invasive PGT, which analyzes DNA from the embryo’s culture medium rather than performing a biopsy, is under active research and shows promise, but it is not yet widely available or validated for routine clinical use. More studies are needed to confirm its accuracy and reliability before it becomes a standard option.
Final Thoughts
Preimplantation genetic testing (PGT) has become an important tool in assisted reproduction, offering the ability to screen embryos for chromosomal abnormalities and specific genetic disorders before implantation. While PGT can improve pregnancy rates per transfer, especially in women over 35 and in experienced centers, it does not guarantee a healthy baby, as not all genetic or developmental issues can be detected at this stage. The technology is rapidly evolving, with non-invasive methods such as analyzing DNA from spent embryo culture media, showing promise for safer and more accessible testing, though these approaches still require further validation before routine use.
Advances in sequencing and bioinformatics are making PGT more comprehensive and efficient, but they also introduce new challenges in interpreting complex genetic data and providing appropriate counseling. Ethical considerations remain central, particularly regarding the fate of embryos found to have abnormalities and the expanding scope of what can be tested. Ultimately, PGT is a valuable option for many couples, especially those at risk for specific genetic conditions, but it is not a universal solution and should be considered within the context of individual needs, clinic expertise, and evolving scientific evidence.
References
-
Evolution of Minimally Invasive and Non-Invasive Preimplantation Genetic Testing: An Overview. Journal of Clinical Medicine, 13. https://doi.org/10.3390/jcm13082160
-
Preimplantation genetic testing for monogenic disorders (PGT-M) offers an alternative strategy to prevent children from being born with hereditary neurological diseases or metabolic diseases dominated by nervous system phenotypes: a retrospective study… Journal of assisted reproduction and genetics. https://doi.org/10.1007/s10815-024-03057-1
-
Preimplantation genetic testing in the current era, a review… Archives of gynecology and obstetrics. https://doi.org/10.1007/s00404-024-07370-z
-
A systematic review and meta-analysis: clinical outcomes of recurrent pregnancy failure resulting from preimplantation genetic testing for aneuploidy. Frontiers in Endocrinology, 14. https://doi.org/10.3389/fendo.2023.1178294
-
Exploring the efficacy and beneficial population of preimplantation genetic testing for aneuploidy start from the oocyte retrieval cycle: a real-world study. Journal of Translational Medicine, 21. https://doi.org/10.1186/s12967-023-04641-2
-
The current clinical applications of preimplantation genetic testing (PGT): acknowledging the limitations of biology and technology. Expert Review of Molecular Diagnostics, 24, 767 - 775. https://doi.org/10.1080/14737159.2024.2390187
-
Genetics in Reproductive Endocrinology and Infertility… Fertility and sterility. https://doi.org/10.1016/j.fertnstert.2023.02.029
-
Preimplantation Genetic Testing for Aneuploidy in In Vitro Fertilization Using Comprehensive Chromosome Screening: A Systematic Review and Meta-Analysis. International Journal of Fertility & Sterility, 18, 185 - 194. https://doi.org/10.22074/IJFS.2023.1996379.1450
-
The impact of reporting low-grade mosaicism in PGT cycles according to the number of cycles with at least one embryo for transfer. Human Reproduction. https://doi.org/10.1093/humrep/dead093.1046
-
Technical and ethical challenges in polygenic embryo selection. **. https://doi.org/10.1101/2024.05.28.24308092
-
A Review of Cost-Effectiveness of Preimplantation Genetic Testing for Aneuploidy. Obstetrical & Gynecological Survey, 80, 169 - 173. https://doi.org/10.1097/OGX.0000000000001373